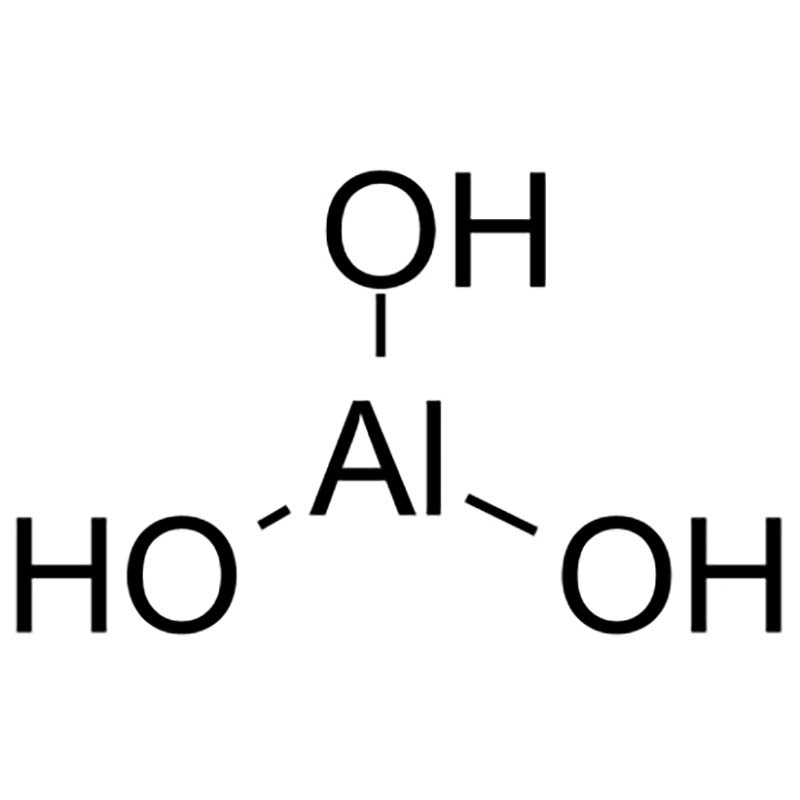
Aluminium hydroxide CAS:21645-51-2
Aluminum hydroxide is a versatile industrial chemical.
Industrial-grade aluminum hydroxide is primarily applied as a filler for plastics and polymer materials, a flame retardant and adhesive in the textile industry, and a packing material for epoxy resins. It also serves as a pigment filler in papermaking and coatings, and is a key raw material for manufacturing aluminum sulfate, alum, aluminum fluoride and sodium aluminate. Additionally, it is used for molecular sieve synthesis, toothpaste filling and glass production, as well as in the preparation of its gel solution and dried gel referenced on Chemicalbook.
Pharmaceutical-grade aluminum hydroxide is widely used as an antacid. It can neutralize gastric acid and protect ulcer surfaces, relieving discomforts caused by hyperacidity and acid reflux. It is indicated for gastric ulcer, duodenal ulcer, reflux esophagitis and upper gastrointestinal bleeding. When combined with calcium supplements and vitamin D, it can also be used to treat neonatal hypocalcemia (tetany).
| Property | Specification |
| Melting Temperature | 300°C |
| Boiling Temperature | 2980C[at 101 325 Pa] |
| Apparent Bulk Density | ~90 g/100 mL |
| Density (at 20°C) | 2.42 g/cm³ |
| Vapor Pressure (at 20°C) | <0.1 hPa |
| Refractive Index | Average: 1.57–1.59 |
| Recommended Storage Conditions | Keep at +5°C to +30°C |
| Aqueous Solubility (at 20°C) | 0.0015 g/L |
| Physical Form | Colloidal suspension |
| Color | White |
| Specific Gravity | 2.42 |
| Odor Profile | Odorless / Flavorless |
| pH (100 g/L slurry in water, 20°C) | 8–9 |
| pH Indicator Transition Range | >7 |
| Water Solubility Characteristic | Practically insoluble |
| Crystal System | Monoclinic |
| Merck Index Reference | 14,342 |
| Solubility Product Constant (pKsp) | pKsp: 32.89 |
| Occupational Exposure Limit (ACGIH) | TWA: 1 mg/m³ |
| Dielectric Constant (Ambient) | 2.2 |
| Stability Profile | Stable under normal conditions; incompatible with strong bases |
| Primary Industrial/Pharmaceutical Uses | Pharmaceutical applications (small-molecule grade) |
| Cosmetic Functional Claims | Skin conditioning agent, emollient, opacifier, humectant, skin protectant, viscosity controller, colorant |
| Cosmetic Ingredient Assessment | Aluminum hydroxide (CAS: 21645-51-2) |
| InChI Identifier | 1S/Al.3H2O/h;3*1H2O/q+3;/p-3 |
| InChIKey | WNROFYMDJYEPJX-UHFFFAOYSA-K |
| SMILES Notation | OAlO |
| Partition Coefficient (LogP, estimated) | -1.380 |
| CAS Registry Number | 21645-51-2 (CAS database reference) |
| EPA Chemical Substance Profile | Aluminum hydroxide (CAS: 21645-51-2) |
Applications of Aluminum Hydroxide
1. Key Chemical Feedstock
Aluminum hydroxide is a widely accessible raw material, thanks to scalable production processes, abundant feedstock supplies, and high purity. Its ability to dissolve readily in both acidic and alkaline media makes it a foundational building block for manufacturing various aluminum salts, including barium aluminate and aluminum sulfate.
2. High-Performance Flame Retardant
As a non-toxic additive, aluminum hydroxide powder serves as a multi-functional filler in plastics, unsaturated polyesters, rubber, and other organic polymers. It provides three core benefits: filling reinforcement, flame retardancy, and smoke suppression.
The flame-retardant mechanism relies on endothermic decomposition: when temperatures rise above 200°C, the compound begins to absorb heat and release bound water molecules, with the decomposition rate peaking near 250°C. This strong endothermic reaction slows the polymer’s temperature increase and decomposition rate, releasing only harmless water vapor without generating toxic byproducts.
3. Advanced Ceramic Precursor
When calcined at high temperatures, aluminum hydroxide converts to aluminum oxide—a material prized for its exceptional thermal stability, mechanical strength, rheological resistance, dielectric properties, and low thermal expansion. These traits make it essential for producing high-performance ceramics. During synthesis, aluminum hydroxide acts as an activator and regulator of crystallization, enabling precise control over phase formation in composite ceramic materials.
4. Water Purification Agent
In aqueous environments, aluminum hydroxide primarily exists as tetrahydroxyaluminate ions ([Al(OH)₄]⁻). It effectively removes toxic heavy metals from wastewater via co-precipitation, followed by simple filtration to purify the water. Its high specific surface area also allows it to adsorb colloids, suspended solids, dyes, and organic contaminants directly onto its surface.
5. Pharmaceutical & Medical Uses
Aluminum hydroxide is a long-standing, safe treatment for gastric conditions due to its acid-neutralizing properties. It also serves as a common vaccine adjuvant: by adsorbing antigens onto its surface, it enables slow, sustained release of the antigen, extending the vaccine’s immunogenic effect and boosting its long-term efficacy.
6. Versatile Catalyst Support
By adjusting reaction parameters such as temperature, concentration, and pH during synthesis, aluminum hydroxide can be tailored to have specific surface areas, pore volumes, pore structures, and crystal forms. These customizable properties make it an effective catalyst carrier for reactions like the hydrogenation of unsaturated carbonyl compounds and fullerene production.
7. Paper & Coating Additive
Thanks to its high whiteness, ultra-fine particle size, well-defined crystal structure, and excellent compatibility with whitening agents, aluminum hydroxide is a valuable additive in paper coatings and resins. It significantly enhances coated paper properties, including whiteness, opacity, surface smoothness, and ink receptivity.